Isobutane vs Butane | Applications in Industry and Trade
Introduction
Among hydrocarbon gases used in the energy and chemical sectors, isobutane and butane are frequently compared. Although both gases have similar properties, their structural differences affect usage areas and commercial value. In this article, the differences between Isobutane and Butane will be examined in detail; advantages in energy, cooling, chemical, and industrial applications, as well as commercial opportunities, will be discussed.
What are Isobutane and Butane?
Butane
Butane (C4H10) is an alkane gas obtained from petroleum and natural gas. It is an important component of liquefied petroleum gas (LPG) mixtures.
Main features:
- Color and odor: Colorless, odorless
- Flammability: High
- Boiling point: Around -0.5°C
- Usage areas: Home heating, industrial fuel, transportation
Isobutane
Isobutane is one of the isomers of butane and, although it has the same chemical formula (C4H10), it shows different properties due to structural differences.
Main features:
- Low boiling point: -11.7°C
- Liquefies under higher pressure
- Preferred in refrigeration and energy sectors
- Important raw material in LPG mixtures and chemical industry
Structural Differences
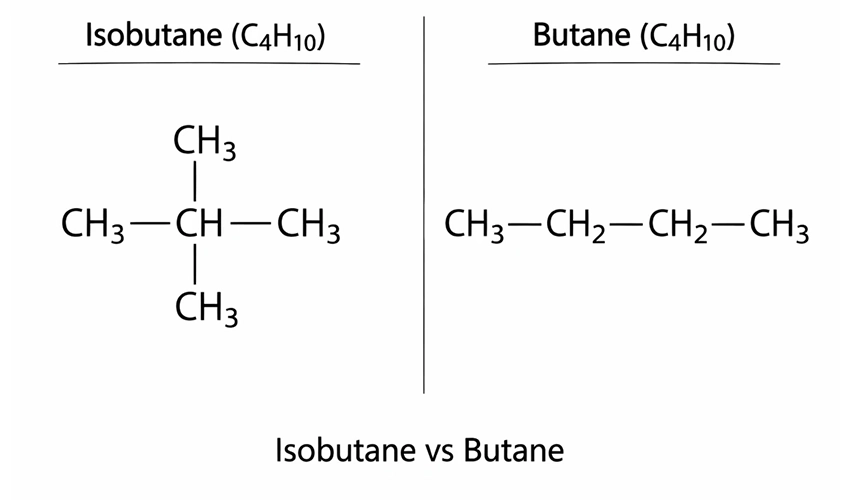
The difference between isobutane and butane primarily comes from their molecular structure.
- Butane: Has a linear structure (n-butane).
- Isobutane: Has a branched structure (2-methylpropane).
Physical and Chemical Property Differences
| Property | Butane | Isobutane |
|———-|——–|———–|
| Molecular Structure | Linear | Branched |
| Boiling Point | -0.5°C | -11.7°C |
| Density | 2.48 kg/m³ | 2.51 kg/m³ |
| Liquefaction Pressure | Lower | Higher |
| Usage Area | Energy and fuel | Refrigeration, energy, chemical |
Usage in the Energy Sector
Both butane and isobutane are widely used in the energy sector, but their usage purposes differ:
Butane:
- In household LPG cylinders
- As industrial fuel
- As an energy source in transportation
Isobutane:
- As a balancing gas in LPG mixtures
- As a combustible gas in power plants
- In low-temperature applications
Differences in Cooling and Chemical Sectors
Cooling
Isobutane is preferred in refrigerants due to its low boiling point and environmentally friendly properties. Butane has limited applications in this area.
Usage areas:
- Household refrigerators
- Air conditioning systems
- Industrial cooling units
Chemical Industry
Isobutane is used as a raw material for the production of isobutylene in the chemical industry. Butane is mainly consumed for energy purposes.
Advantages:
- Isobutane: Lower toxicity, eco-friendly, and efficient use
- Butane: Economical energy source
Logistics and Storage Differences
- Butane: Can be stored and transported at lower pressure.
- Isobutane: Requires higher pressure but provides advantages for cooling and chemical use.
Environmental Impacts
- Butane: CO2 emissions are higher; environmental impact is limited to energy use.
- Isobutane: Ozone-friendly, less harmful to the environment, and lower greenhouse effect compared to HFC gases.
Commercial Advantages
- Isobutane carries higher value for commercial LPG producers and chemical companies.
- Butane provides cost advantage in the energy sector.
- Using isobutane adds extra value for companies in terms of eco-friendly image and sustainable production.
Conclusion
In summary, the difference between Isobutane and Butane is evident in molecular structure, physical properties, usage areas, and environmental impacts.
- Butane: Used for energy purposes, economical, and has a linear structure.
- Isobutane: Preferred in refrigeration, chemical, and special energy applications; eco-friendly and efficient.
Choosing the right gas is critical for commercial companies in terms of cost, efficiency, and environmental compliance. Therefore, awareness of the differences between Isobutane and Butane is a strategic advantage for industry professionals.